Published on September 20, 2020 by Kenneth D. Steinsapir, M.D.
This is a perspective that I published on the Realself.com website in 2011. For whatever reason, Realself.com has decided to no longer host this type of doctor generated content. Because this article features significant information not available elsewhere, I am republishing it on my blog so readers have access to this information. I do not perform iris color surgery. However, if you have had this type of surgery and are in trouble, please feel free to contact me, I will assist you to help you find the medical care you need close to home. I know that if you have had this surgery, you have agreed to a nondisclosure agreement. These agreements are not valid. Don’t let this stop you from getting the help you need. Act to save your vision. People have gone blind from this type of surgery.
If you are reading this article, perhaps you are doing research on eye color surgery. Please read this article very carefully so you might avoid this surgery and the severe complications that are associated with this surgery. The method has been marketed on-line under the name of NewColorIris®.
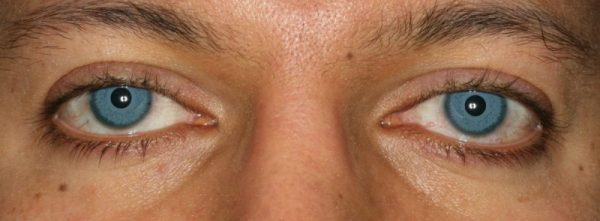
A second company claims to be marketing an improved iris color implant also for the purpose of cosmetically changing the eye color and for other medical reasons under the trademark Brightocular. Individuals have informed me that these services were related (read Anita’s letter below). I carefully researched this issue at the time I originally uploaded this article. Online posters have indicated that they have contacted the New Color Iris website and have been referred to Brightocular. However, I could not confirm the relationship between Brightocular and New Color Iris so I opined that it appeared the two were related. I felt that this was a fair comment on a matter of public interest. However I was not definitive based on the information I gathered from my research.
Threats of Legal Action
Attorney Kevin J. Abruzese of Mineola, New York contacted me regarding this matter. In his certified letter dated August 16, 2011, he indicated that he represented Stellar Devices LLC “the owner of the trademark rights for the term Brightocular.” Referencing my original article, he claimed that:
A review of the article reveals that you have made defamatory comments regarding Brightocular. Specifically, by comparing it to NewColorIris an unrelated independent company. Please be advised that the product marketed under the name Newiris exists under a separate and distinctly different patent owned by Dr. Kahn in Panama and is not structured exactly the same as the product marketed under the name Brightocular. Brightocular is in no way associated with NewColorIris; New Iris or any other company that markets and/or makes products intended to change the color of the iris. In addition, Brightocular significantly decreases certain risks you speak of and thus, you are providing false, misleading and disparaging information by stating that the Brightocular product owned by StellarDevices LLC. in the United States is the same as the product in Panama in reckless disregard of the truth.
Further, Stellar Devices LLC is currently working on obtaining CE marking so implants can be used all over Europe for medical reasons such as coloboma, aniridia and ocular albinism. Moreover, Stellar Devices LLC is presently working with Minnesota Eye Consultants located at [Address withheld] to obtain FDA compassionate approval for a patient who had aniridia. Thus Stellar Devices LLC’s implant is not intended to be solely used for cosmetic reasons.
This defamatory content has placed Brightocular in a false light with the general public, which may have caused Stellar Devices LLC. damages.
The letter goes on to demand that I take immediate action to remove any and all defamatory content from any and all websites, etc. Reviewing what was written, I believe that my statement was clearly an accurate assessment based on research and represented a fair comment on a matter of public interest. I don’t agree at all with Mr. Abruzzese’s characterization of this article. I also question some of his claims. He stated “Brightocular is in no way associated with NewColorIris.” I wonder if he meant this in some type of narrow legal sense? The reason why I question the accuracy of his statement has to do with the fact that the trademark Brightocular is not registered to Stellar Devices LLC as he stated in his letter.
On August 23, 2011, I performed a registration search on the trade name BrightOcular. This mark is a properly registered trademark with the United States Patent and Trademark Office as Serial number 77962791 and Registration number 3949067, originally filled on March 18, 2010, and granted registration on April 19, 2011. The company to which the trademark is registered however is not Stellar Devices LLC. No, according to the United States Patent and Trademark Office, the tradename is registered to New Color Iris, LLC located at a Brooklyn street address. Interesting a Google search on Stellar Devices, LLC revealed that Stellar Devices, LLC and NewColoriris, LLC shared the same address. Are these actually independent companies as claimed by Mr. Abruzzese? I believe that reasonable people can reach their own conclusions about this.
Claims of a New Design
However, it is important to appreciate that Mr Abruzzese claims that the BrightOcular implant is different than the New Color Iris implant. It is fair that this company have an opportunity to prove themselves with appropriate clinical testing and in peer-reviewed publications. There are significant potential medical reasons for anterior chamber iris implants. Provided clinical testing demonstrates a reasonable track record of safety, this device may very well have a potential medical role based on an appropriate risk/benefit ratio. However, it is my opinion that there are enough questions regarding the safety of this type of device that, without a track record of clinical safety, I can not recommend that individuals have an iris implant for the purpose of cosmetically changing their eye color for reasons I lay out below. It is essential to recognize that there are absolutely no published clinical papers on the Brightocular implant. We accept and welcome that it may be an improved design over prior products. However, again in my opinion, this is not the basis for deciding to have this device inserted into a healthy eye without through clinical testing and a proven track record of safety. So far this is lacking. I have called upon Stellar Devices, LLC to voluntarily withdraw their device for the purpose of cosmetically changing eye color until more is known clinical about this device in the form of peer-reviewed, published studies.
Does flying to Panama or Turkey really make sense?
On my website (lidlift.com), I have posted an article about iris color surgery a couple of year ago. The surgery is a procedure that changes the color of the eye by inserting a colored silicone disc inside the eye in front of the iris. Before you rush out looking for this surgery, please carefully read the rest of this article. I was reminded of my website post by a very nice individual who emailed me about her terrible experience with this surgery. I have her permission to quote her email.
Dear Dr. Steinsapir
I was just reading the entry in regard to iris implants for the sole purpose of eye color change. I was one of the unfortunate patients who traveled to Panama and had Dr. Delarey Kahn perform the implant surgery on me. I had the implants inserted in October of 2008 and I am now having to pay thousands of dollars to get the devices removed and hope that my vision does not suffer any more than it already has. It is no coincidence that this Doctor’s last name is Kahn (Con). I have made several attempts to reach him since I started having problems, but to no avail. I had the first implant removed yesterday and I will have the other on removed on February 1st. After I first had the implants, I made an appearance on their behalf on Inside Edition. At this point I am just doing the best I can t warn people of the complications that may occur no matter how safe they make it sound. New Color Iris was the first company to market this procedure and now they are referring people to a company called Bright Ocular, which is performed in Instanbul. It sounds exactly like New Color Iris. If there is no FDA approval, there is a reason. I had to find this out the hard way.
Sincerely,
Anita
[Author’s note: As discussed above, representatives of the Brightocular implant claim that Brightocular is unrelated to New Color Iris.]
There are now numerous scientific papers reporting permanent damage and loss of vision as a direct result of iris color surgery. Authors George, Tsai, and Loewen (Am J Ophthalmol 2011;151:872–875) recently reported the case of a 25 year old woman who had implantation of the colored iris implants to change the color of her eyes in Panama. Within a week of her surgery, she developed blurred vision, red eye, light sensitivity, and glaucoma. She was advised to have the implants removed but did not follow this advice until 16 months later. By this time, she suffered severe damage to the eyes from a process called neovascular glaucoma as a result of the chronic inflammation from the implants. Despite heroic efforts by ophthalmologists in the United States, she lost all usable vision in the right eye, and was left with a tiny island of poor vision in the left eye. This is not the only case of individuals suffering from complications from this surgery.
The device was invented by a Panamanian ophthalmologist Delary Kahn, M.D. According to his promotional materials, he invented the implant to help the Kuna Indians of Panama who suffer a high incidence of ocular albinism. However, the implant is primarily promoted as a cosmetic treatment to permanently change eye color. Internet marketing material states:
“Now you can Permanently Change Your Eye Color to a beautiful and natural looking Blue or Green you dreamed of with a safe and painless cosmetic surgery; never use contact lenses again…”(newcoloriris.com/index.html accessed June 14, 2011)
However, this surgery is anything but safe. Authors Anderson, Grippo, Sbeity and Ritch writing in Acta Ophthalmologica (Vol 88, no 6 published online: 2 Jun 2009) reported two patients who suffered multiple complications for their NewColorIris implants. Both had to have the implants removed. One required advanced glaucoma surgery and the other may need corneal surgery from complications arising from the implants. Another set of investigators, Arthur, Wright, et al (AJO 2009;148:790-793, 2009) reported on a 29 year old who developed uveitits (internal eye inflammation), glaucoma, and hyphema (bleeding inside the eye) and corneal decompensation. He required removal of the implants and to save vision in one of the eyes, advanced glaucoma surgery was required.
How common are these complications? It is impossible to know. The inventor of the device has made very little effort to disseminate his results in peer-reviewed literature. The reported complications are completely foreseeable due to the nature of the implant and where it is placed in the eye. It is very hard to see how this procedure is safe for a healthy eye. Having this surgery to change the color of your eyes places you at risk of going blind. If you are even remotely considering this surgery, this statement should stop you in your tracks. There is absolutely no evidence that this surgery is safe. You are far safer just using colored contact lens and being followed carefully by your eye care professional.
The device lacks FDA approval and given the severity of the complications from the device, it is improbable that approval would ever be granted for cosmetic use in the United States. The presentations to date by Dr. Kahn and his colleagues only include 12 patients, although website materials imply that more have been treated.
Yet another report on this implant was published by Thiagalingam et al (Thiagaligam, et al. Complications of cosmetic iris implants. J Cataract Refract Surg 2008; 34:1222-1224). These authors report the case of a 19-year old man who presented 2 weeks after implantation of colored iris implants with reduced vision, inflammation inside the eye, and glaucoma. Both color iris implant were ultimately removed to treat the inflammation. The authors of this report found that this man had lost a larger percentage of endothelial cells and had early cataract changes thought to be directly related to the placement of the colored iris implants.
There is a long history of anterior chamber implants to correct vision after cataract surgery and more recently so-called phakic intraocular lenses have been introduced to correct vision for individuals who are not candidates for refractive surgery on the cornea, like LASIK. Unlike the colored iris implant, a number of these intraocular implants are FDA approved. Further, clinical experience with these phakic intraocular lenses are well described in peer-reviewed papers. Even these FDA approved devices are controversial. The key issue is related to the balance between risk and benefits. When the benefits are outweighed by the risks, responsible surgeons will strongly discourage the treatment.
We now have enough reports of visual loss and visually threatening complications prompting the surgical removal of these iris color implants to strongly advise the public not to have this procedure for cosmetic purposes.
If you don’t like your eye color see your contact lens specialist for colored contacts. However, save your vision. Don’t have iris color surgery for this purpose. AskMen.com called it right in reviewing the technology: “Normal people don’t permanently change their eye color. Crazy people permanently change their eye color.” If you still feel compelled to have eye color surgery, see you personal general ophthalmologist, and ask them what they think of the idea because this person will be responsible for saving your vision if your are foolish enough to ignore this warning.

